We are experiencing a very high volume of calls and messages and ask for your patience. We will answer your portal messages within 48 hours.
We are experiencing a very high volume of calls and messages and ask for your patience. We will answer your portal messages within 48 hours.
.jpg)
.jpg?width=300&height=168&name=My%20project-1%20(89).jpg) The treatment of male factor infertility was revolutionized in 1992 when Palermo and co-workers introduced intracytoplasmic sperm injection (ICSI). With ICSI, embryologists use a micromanipulator to inject sperm directly into the egg (that has been retrieved as part of an IVF cycle). It was now possible for men with severe male factor infertility to father a child. ICSI can be used even in cases where the wife produces more eggs than the husband produces sperm!
The treatment of male factor infertility was revolutionized in 1992 when Palermo and co-workers introduced intracytoplasmic sperm injection (ICSI). With ICSI, embryologists use a micromanipulator to inject sperm directly into the egg (that has been retrieved as part of an IVF cycle). It was now possible for men with severe male factor infertility to father a child. ICSI can be used even in cases where the wife produces more eggs than the husband produces sperm!
Using high magnification, an oval-appearing motile sperm is selected for ICSI. When a sperm is motile-- moving and swimming-- it indicates that it is viable and therefore is capable of fertilizing the egg.
But, what is to be done when there are no motile sperm? This condition is called “absolute asthenozoospermia” and affects 1 in 5000 men. Ortega et al. discussed this issue in the journal Human Reproduction Update. Here is a summary of their findings.
The two main causes of absolute azoospermia in ejaculated sperm are:
Each one of these entities needs to be diagnosed and treated appropriately.
When testicular sperm aspiration (TESA) is done, that sperm is usually immotile due to metabolic sperm immaturity or attachment to Sertoli cells.
To diagnose absolute azoospermia, the sperm sample should be produced after a standard abstinence period of 3 days. Care should be taken to examine the sample at 37 degrees C. It is extremely important to assess a second sample as sometimes, the second sample can contain motile sperm (even when the first one does not). Any infection present must be treated with antibiotics.
If no cause is found or no treatment is available, the only option is to carefully select sperm that can be used for ICSI.
If all sperm are dead (necrozoospermia), there is no point in using ejaculated sperm. In this situation, testicular sperm aspiration (TESA) may help. The procedure is preferably done on the day before egg retrieval. There have been cases where sperm of testicular origin started moving after overnight incubation in culture medium.
Several techniques have been described to assess viability in immotile sperm. One is the hypo-osmotic swelling (HOS) test and there is a mechanical touch technique. In the HOS test, the sperm are cultured in a solution that will permeate through the membrane of (only) a viable sperm. This makes the tail curl, making it possible to identify the sperm as being viable.
In the mechanical touch technique, the sperm is touched with the ICSI pipette. The test is considered to be positive if the tail bends and recovers to the initial position. Other techniques include exposure to pentoxifylline, using a non-contact 1.48 mm diode laser to induce sperm tail curling, and birefringence-polarization microscopy. There are no data indicating that one technique is better than the others.
The following figure shows steps that should be followed when treating a patient with absolute azoospermia:
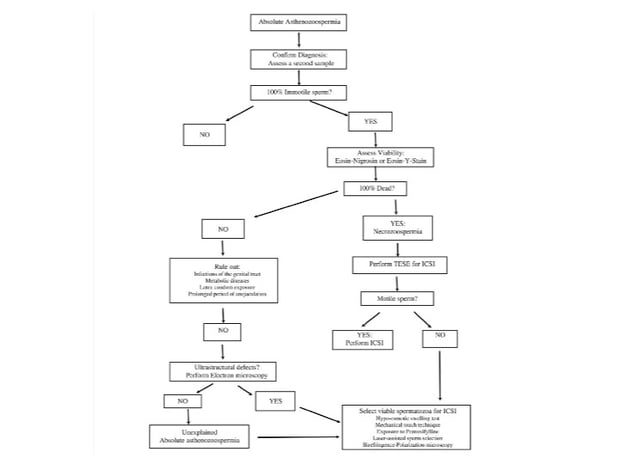
Success rates will vary depending on the ability to find viable sperm. There have been several small studies where the pregnancy rates vary from 3 – 76.4%.
Absolute azoospermia has a poor prognosis, especially when no viable sperm are detected. A thorough work up is indicated to diagnose and treat correctable factors.
We can help you assess male-factor infertility issues and explore your options for getting pregnant. Make an appointment at one of InVia’s four Chicago fertility clinics today.

Entire Website © 2003 - 2020
Karande and Associates d/b/a InVia
Fertility Specialists
